New cancer therapy being studied at UAB could one day offer complete radiation treatment in milliseconds
A full course of radiation therapy in milliseconds rather than weeks: That’s the promise of flash therapy.
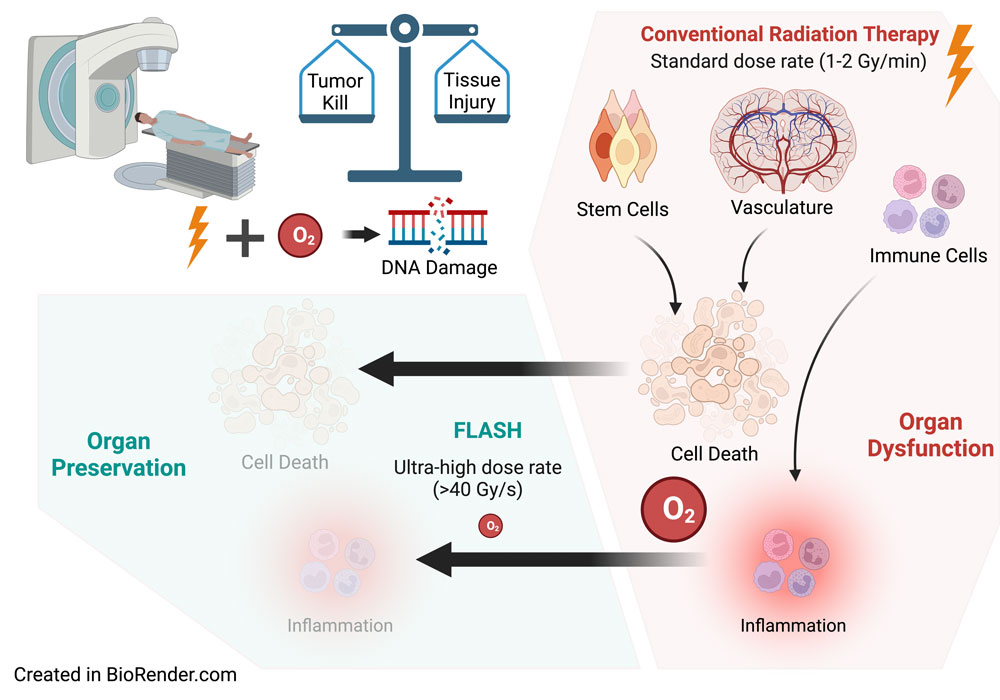
For a typical cancer patient, radiation therapy is a six-week slog. After each week of five daily treatments, many patients experience an accumulation of side effects. Following the full treatment course, they spend the next several weeks trying to recover from the fatigue, swelling, tenderness and other side effects. But an experimental treatment called flash radiation therapy may be able to get the same anti-cancer effect in a single visit, with one burst of radiation less than a second long — and with fewer side effects. (See an illustration of the process in the video and graphic below.)
This sounds too good to be true. After all, the whole reason for splitting up the radiation dose over several weeks, a process known as fractionation, is to give the body a chance to recover from the DNA damage (and subsequent inflammation and organ dysfunction) that radiation causes. But, paradoxically, concentrating that dose in one ultra-high, ultra-fast burst seems to have the same deadly effect on the tumor as normal radiation, while sparing healthy cells and many of those side effects.
How fast is flash? Radiation oncologists measure the amount of radiation absorbed by their patients in units called Gray. “A standard course of radiation is roughly 60 Gray given over six weeks,” said Christopher Willey, M.D., Ph.D., Hale-Stephens ROAR Endowed Professor for Distinctive Radiation Research and associate vice chair of Translational Research in the Department of Radiation Oncology at the UAB Heersink School of Medicine. “With flash, you would get an equivalent dose in less than a second. And there is evidence that, when you give radiation that fast, normal cells are harmed less.”
Although the flash effect was noted more than a half century ago, a landmark paper from European scientists in 2014 spurred the current interest in the field. Within five years, flash had moved to human studies, with the report of the first patient treated using flash radiation therapy in 2019. The first human clinical trial of flash therapy, published last year in JAMA Oncology, included 10 patients with bone metastases. And more trials are coming soon, says Willey, who is also a scientist at the UAB O’Neal Comprehensive Cancer Center. UAB is one of 30 institutions worldwide participating in the FlashForward Consortium with Varian.
“A lot of excitement around this research”

In addition to the successful human clinical trial reported last year, there have been several veterinary trials on pet cats and dogs with tumors that have shown “pretty remarkable” results, Willey said. “There has been a lot of excitement around this research, but people are still trying to figure out how it really works.” There are lots of theories about that, Willey added, “many of them relating to oxygen or the immune system.” For instance, the high dose of radiation aimed at the tumor may temporarily use up all the oxygen in the area, sparing healthy tissues from the DNA damage that radiation brings. There is another key question to be answered as well: Which types of cancer or locations in the body are most amenable to flash therapy?
Willey and UAB colleague Lewis Shi, M.D., Ph.D., Koikos-Petelos-Jones-Bragg ROAR Endowed Professor for Cancer Research at UAB and director of Radiobiology in the Department of Radiation Oncology, have each received grants to study the mechanisms of flash radiation in their labs. Willey is studying flash therapy in brain tumors (glioblastomas) in both animal models and in cell culture, while Shi is doing the same for melanoma. “We are both trying to investigate this flash effect,” Willey said. “Most flash studies have been done in animals, but in vitro [cell culture studies] gives us the opportunity to use more tests and work at higher speed.”
Reaching the radiation doses necessary for flash “is not easy,” Willey said. “Most existing studies have been done with electrons from standard linear accelerators” that have been modified to increase the output, he explained.
The data that he and Shi are generating “will probably contribute to the mass of data being produced worldwide, particularly in this consortium,” Willey said. The first human clinical trial was in patients with bone metastases, which are relatively simple tumor types in easily accessible locations, Willey points out. “Now we need to prove that it is safe in various parts of the body. That is the next challenge.”